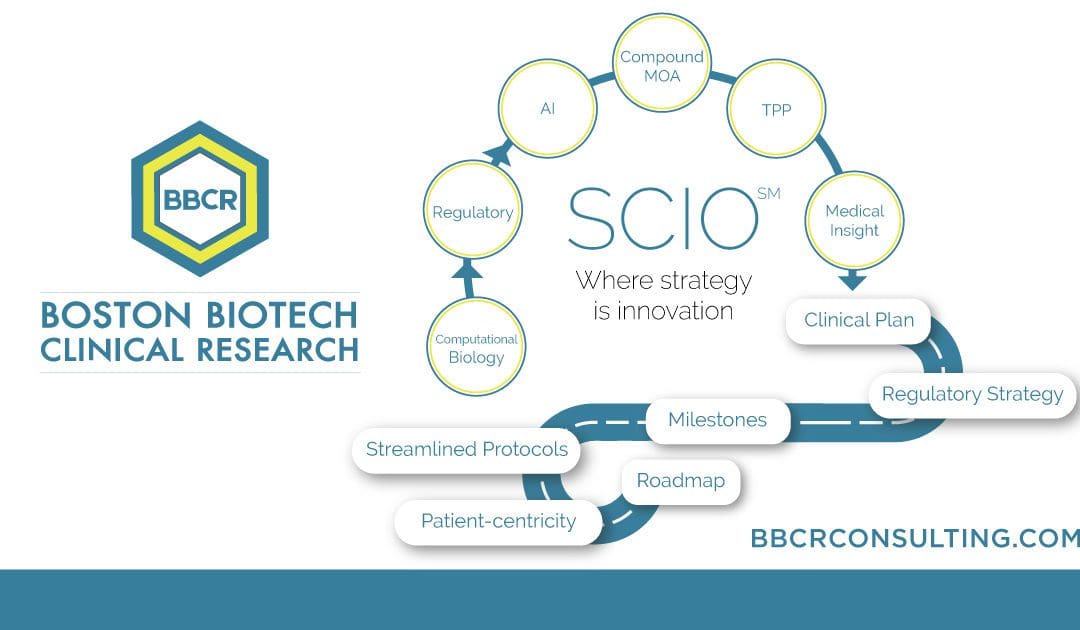
Innovation in clinical research is a long-term need due to several factors, including the high failure rate and skyrocketing costs. Several factors contribute to the skyrocketing cost of clinical trials. Integration of advanced technologies into drug development. While these technologies offer valuable insights, they incur additional training, data analysis, and interpretation costs. Complexity of study […]
