Strategic Clinical Innovation Organization (SCIO SM)
SCIO SM Path to Market Approval
The Strategic Clinical Innovation Organization SM (SCIO) method is explicitly designed to help pharmaceutical innovators address their concerns and maneuver around evolving challenges. SCIO SM identifies time and cost efficiencies and relief from risk management on their journey to market approval.
Why the SCIO SM Method
The SCIO SM method aims to learn and predict so better decisions can be made for a successful drug opportunity.
It learns the potential of each preclinical asset and predicts the level of certainty for each strategic option.
Today’s technological molecules require innovation in the development process. We cannot keep using yesterday’s assumptions for tomorrow’s drugs.
-
- Evidence from marketed products proves that identical molecules developed by different sponsors generated very different drug opportunities.
- Data shows that evaluation of options and early strategy (from indication selection and validation into phase I-II trial) lead to a competitive advantage that a late development redirection of optionality and decisions cannot replace.
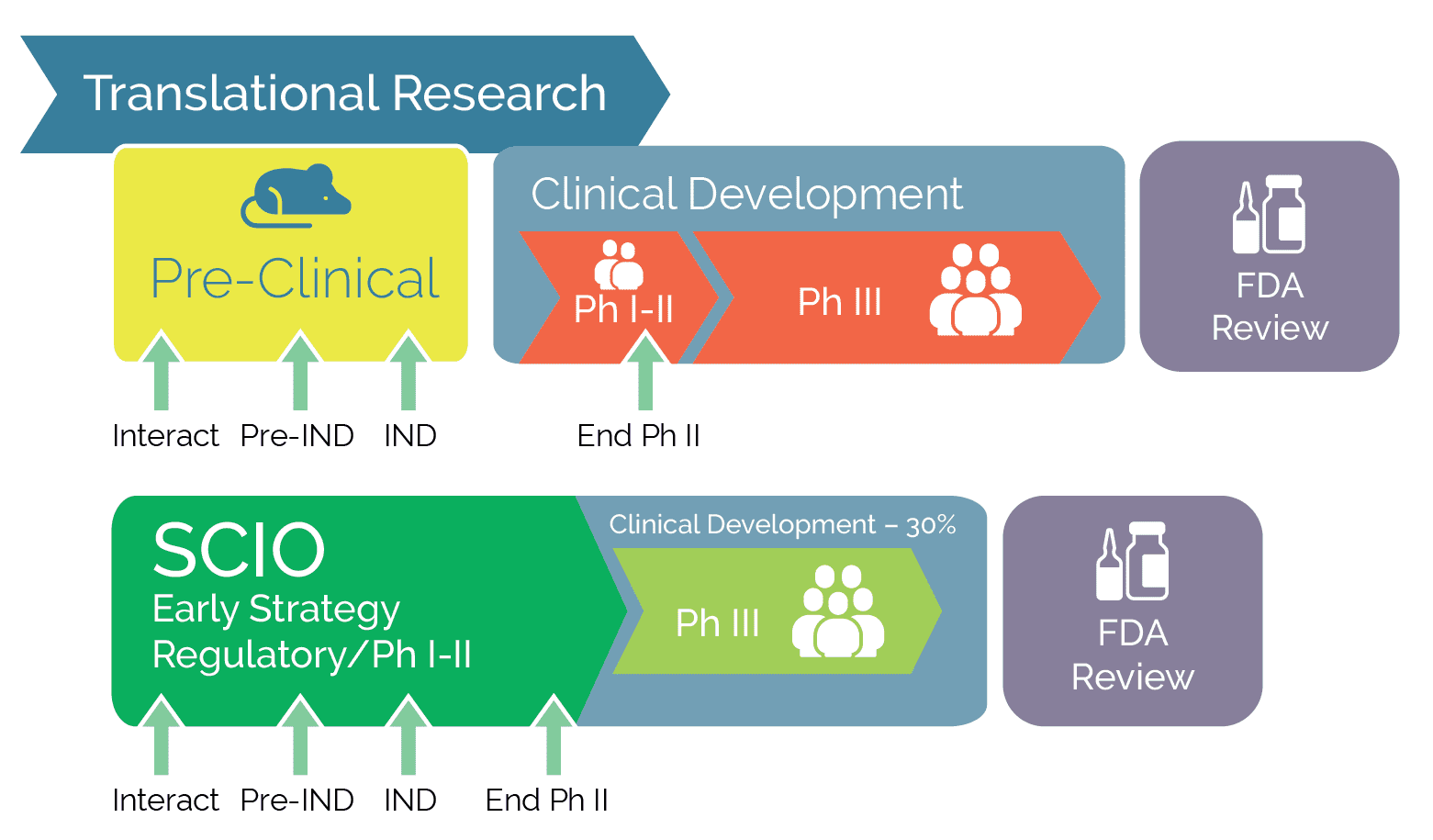
An early-phase strategy improves productivity and the path to market approval
The SCIO SM method enhances and drives strategy-based early-phase decisions and manages risk within the early phases of clinical research, thus increasing productivity.
An early phase strategy considers alternative development paths, indications prioritization, patients’ needs, and regulatory risks.

The SCIO SM Method Deliverables
An early-phase strategy improves productivity and the path to market approval.
For Sponsors, this approach is critical to predicting the level of certainty needed for quicker, less expensive, and a higher revenue path to market approval with lower regulatory risk.
For VCs, it is critical for understanding a product’s qualities and predicting the certainty of potential for market approval. Even if the team is stellar, the odds of market approval are very low without knowing the quality and potential of the opportunity.
Flexible Road-Map
Successful clinical development for drug and medical device companies (small and large) requires an early strategy with a flexible roadmap to make it actionable and sustainable.
Robust Clinical Data
A customized strategy and roadmap allow for robust clinical data generation and informed decision-making.
Clear Goals
Setting clear goals is essential to managing risk and pre-empting challenges. The BBCR team adopts innovative solutions and strategies thanks to scientific, medical, regulatory, and clinical research experience.
Cost-Effective Trials
A customized clinical/regulatory strategy, roadmap, and continuity plan pave the way for cost-effective trials.
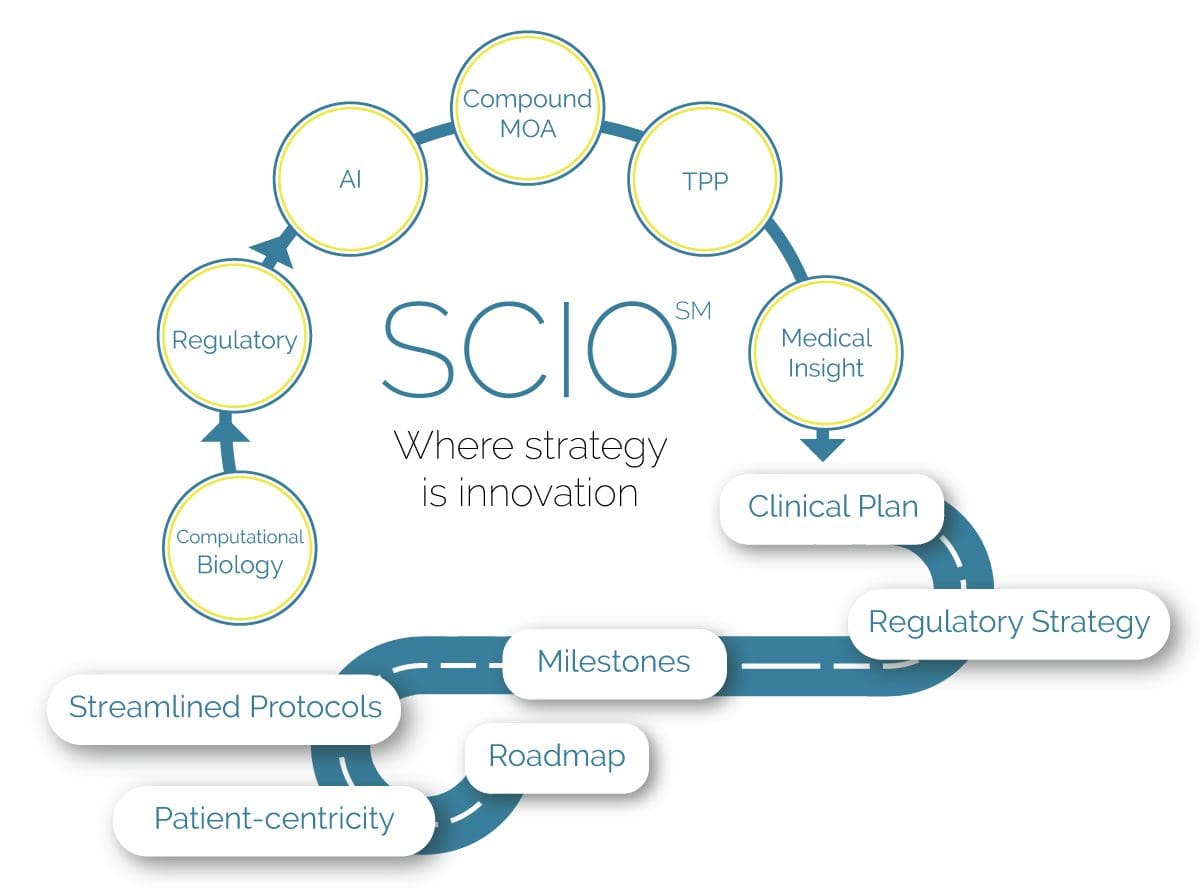
SCIO SM Advantages
- Accelerate Patient Recruitment
- Reduce Patient Number
- Reduce Clinical Development Time
- Reduce Trial Monitoring Time
- Increase Patient Retention
- Facilitate Decision Making
- Increase Data Quality
The FDA has been calling for a smarter, more innovative process for market approval, and SCIO SM is the integrative, multidisciplinary approach to deliver it.

We Create a Roadmap Customized for your Product
The BBCR team is armed with extensive clinical, regulatory, and industry experience that we use to create a product-specific clinical/regulatory strategy as part of the IND or IDE before tactical clinical activities start. We aim to transform the transition stage between pre-clinical and clinical endpoints with plans that, at a minimum, reduce costs to benefit patients.
- Design-centric trials
- Patient-centric approach
- Simplified trials
- Streamlined development plan
- Cost efficiency
- Time efficiency
- Risk management
