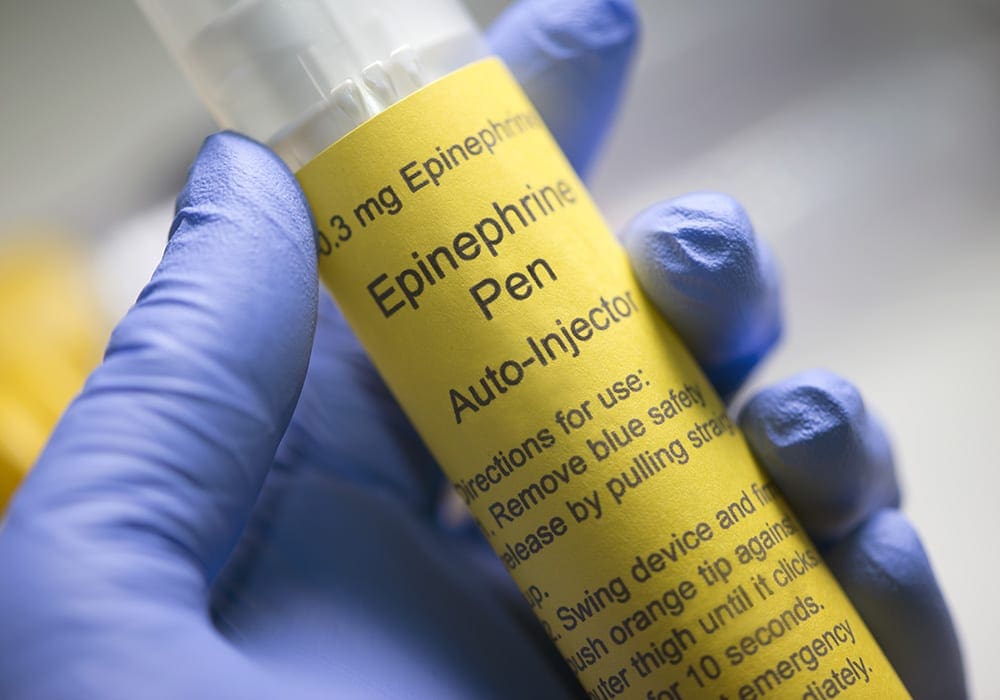
Teva received approval to market generic versions of Mylan’s epinephrine auto-injector for emergency treatment of allergic reactions for both adults and children in August 2018 after several years of delays in getting the generics approved. Teva Pharmaceutical EpiPen Jr. (epinephrine) auto-injector 0.15mg is market at a wholesale cost of $300 per two-pack. This is the first generic […]
