How we can help The BBCR team provides knowledge in cell, biologics and gene therapies BBCR addresses sponsors’ questions in the ever-changing regulatory environment We offer clinical strategy for orphan diseases and precision medicine RWE can be used to build orphan diseases evidence to support regulatory decisions BBCR serves small and medium sized Biotech Companies, […]
Tagged: cell therapy
The BBCR mission is to customize strategies, simplify clinical research, design cost-effective trials, streamline protocols, and create a regulatory roadmap. Learn more about our services by visiting bbcrconsulting.com
September 20th, 2022 | cell therapyTrial for Duchenne muscular dystrophy cell therapy.
January 27th, 2022 | cell therapy
Duchenne Muscular Dystrophy is the most common fatal genetic disorder to affect children around the world. The first patient, a six year old boy, received, in November 2021, an infusion of DT-DEC01, a novel cell therapy containing Dystrophin Expressing Chimeric Cells (DEC), in Phase I pilot clinical study for the treatment of Duchenne muscular dystrophy (DMD). […]
Drug Development for Rare Duchenne Muscular Dystrophy (DMD)
April 20th, 2021 | cell therapy
Progress and limitation of therapeutic approaches targeting DMD By: Dr. Maria Niu Duchenne muscular dystrophy (DMD) is an X—a chromosome-linked recessive disorder caused by mutations in the gene coding muscle cytoskeletal protein dystrophin. Because of the X-linked inheritance, nearly 1 in 3,500 males are affected worldwide, while females occasionally develop symptoms. There is no curative […]
Cell and gene advanced therapy in the Battle for Lysosomal Storage Disease
April 8th, 2021 | cell therapy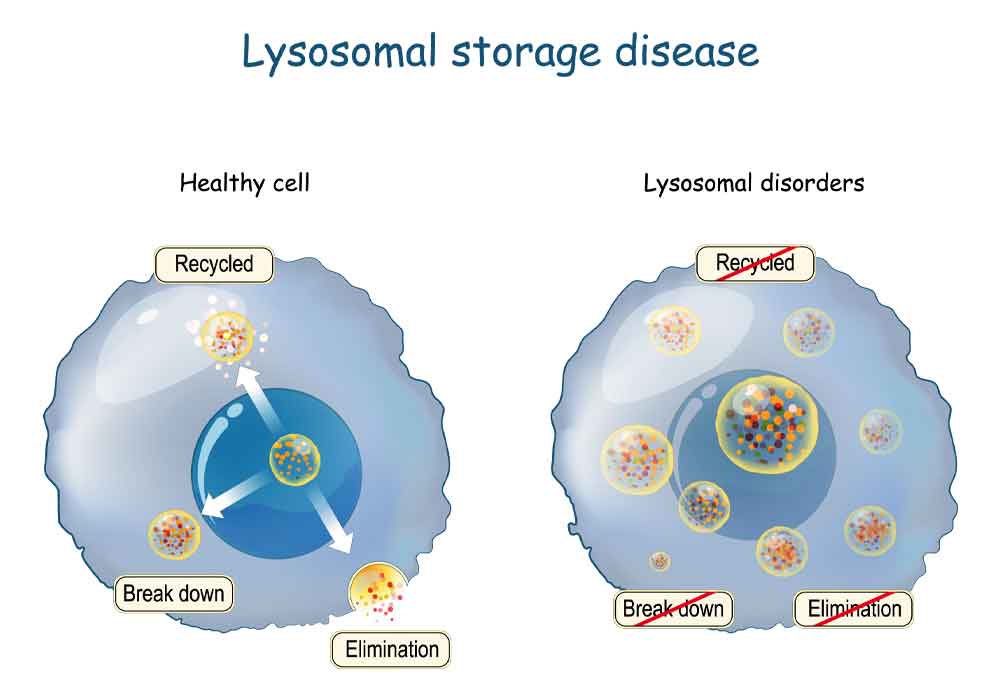
Emerging cell and gene therapy may offer sustained long-term correction for LSD patients Dr. Maria Niu Lysosomal storage diseases (LSDs) are rare inherited metabolic diseases and characterized by the accumulation of substrates in excess in various organs’ cells due to lysosomes’ defective functioning. The combined incidence of LSDs is between 1 in 5000 to 1 […]
